- Who came up with the first version of the Periodic Table that we use today?
- Rimski Korsakov
- Gregor Mendel
- Dimitri Mendeléev
- Mendeléev’s table was arranged in order of…
- Atomic mass
- Atomic number
- The modern periodic table is arranged in order of…
- Atomic mass
- Atomic number
- The columns in the periodic table are called…
- Groups
- Periods
- Verticals
- Elements in the same group have…
(choose the best answer)- similar physical properties
- similar chemical properties
- similar appearance
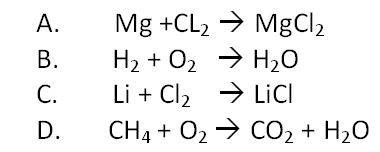 Which of these equations is balanced?
Which of these equations is balanced?- -
- -
- -
- -
 What is this element?
What is this element?- Krypton
- Phosphorus
- Klaxon
- Potassium
 What is the atomic number of this element?
What is the atomic number of this element?- 39
- 39 + 19
- 39 - 19
- 19
 How many protons are there in the nucleus of this element?
How many protons are there in the nucleus of this element?- 39
- 39 + 19
- 39 - 19
- 19
 How many electrons are there around the nucleus of this element?
How many electrons are there around the nucleus of this element?- 39
- 39 + 19
- 39 - 19
- 19
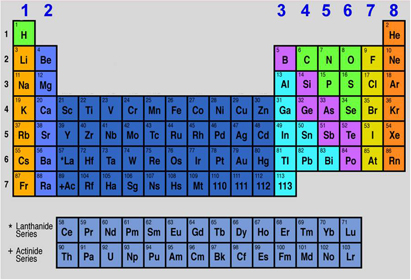 Which group number are the halogens?
Which group number are the halogens?- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
 Which group number are the alkali metals?
Which group number are the alkali metals?- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
 Which group number are the noble gases?
Which group number are the noble gases?- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
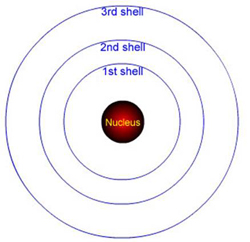 How many electrons can the 1st shell hold?
How many electrons can the 1st shell hold?- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
 How many electrons can the 2nd shell hold?
How many electrons can the 2nd shell hold?- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- Electrons can only orbit in these fixed shells, and can jump between them.
This explains… (choose the best answer)- why elements produce distinctive colours when heated
- why white light is a mixture of colours
- why we see line spectra
 How many electrons do the alkali metals have in their outer shell?
How many electrons do the alkali metals have in their outer shell?- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
 How many electrons do the halogens have in their outer shell?
How many electrons do the halogens have in their outer shell?- 1
- 2
- 3
- 4
- 5
- 6
- 7
- 8
- Metals tend to give electrons away when they form bonds.
- True
- False
- Metals form positive ions when they form bonds
- True
- False
- Ionic bonding is about…
- atoms sharing electrons with their neighbours
- metal atoms giving electrons away and non-metals gaining electrons
- metal atoms gaining electrons and non-metals giving electrons away